United States In Vitro Diagnostics Market Size Growth, Key Players & Latest Industry Trends Report 2034
United States In Vitro Diagnostics Market Size, Share, Trends and Forecast by Test Type, Product, Usability, Application, End User, and Region, 2026-2034
Market Overview
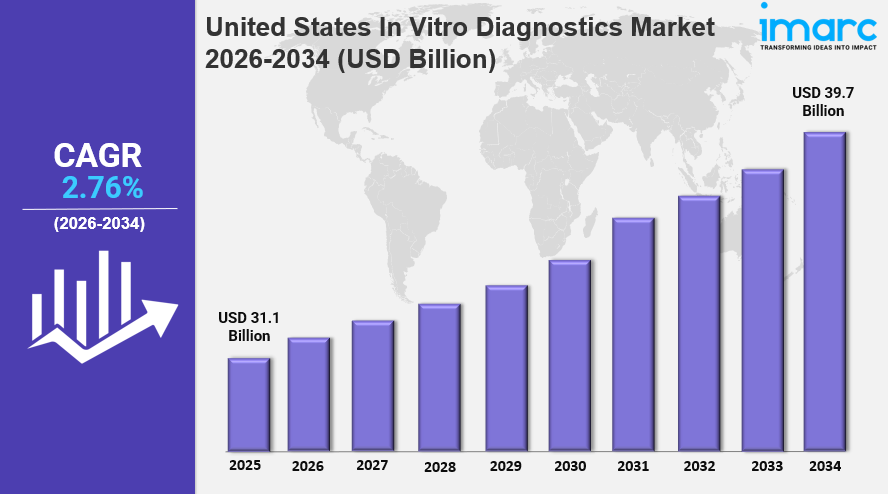
The United States In Vitro Diagnostics Market was valued at USD 31.1 Billion in 2025 and is projected to reach USD 39.7 Billion by 2034, growing at a CAGR of 2.76% during 2026-2034. This growth is fueled by the rising prevalence of chronic diseases, technological advancements in diagnostic tools like molecular diagnostics, and increasing demand for personalized medicine. Supportive government initiatives and integration of AI and machine learning in diagnostics further augment market expansion.
Study Assumption Years
- Base Year: 2025
- Historical Year/Period: 2020-2025
- Forecast Year/Period: 2026-2034
United States In Vitro Diagnostics Market Key Takeaways
- Current Market Size: USD 31.1 Billion in 2025
- CAGR: 2.76%
- Forecast Period: 2026-2034
- The market growth is driven by the rising prevalence of chronic diseases such as diabetes, cardiovascular diseases, and cancer.
- Technological innovations like PCR and next-generation sequencing are enhancing diagnostic sensitivity and specificity.
- Government actions include FDA regulations that mandate compliance with medical device standards for in vitro diagnostics.
- AI and machine learning improve diagnostic accuracy, speed, and scalability.
- The demand for personalized medicine drives the need for specialized diagnostic equipment.
Sample Request Link: https://www.imarcgroup.com/united-states-in-vitro-diagnostics-market/requestsample
Market Growth Factors
The United States in vitro diagnostics market growth is chiefly propelled by the increasing prevalence of chronic diseases, including diabetes, cardiovascular diseases, and cancer, predominantly affecting the aging population. This has surged the demand for accurate and early diagnostic tools, enabling instant disease identification and treatment. These chronic disorders require advanced diagnostic tools to improve patient outcomes, fueling market demand.
Technological advancements, particularly in molecular diagnostics, such as polymerase chain reaction (PCR) and next-generation sequencing (NGS), have significantly improved diagnostic capabilities by delivering highly sensitive and specific results. For example, Tempus received FDA approval in April 2024 for its xT CDx, an NGS-based diagnostic profiling 648 genes in solid tumors, facilitating personalized cancer treatments. Such innovations are driving adoption across hospitals, clinics, and laboratories nationwide.
Government initiatives also significantly influence market growth. The FDA finalization in May 2024 of regulations classifying in vitro diagnostics (IVDs), including laboratory-developed tests (LDTs), as medical devices, enforces compliance with medical device reporting, quality systems, and premarket reviews. Additionally, incorporation of AI and machine learning technologies enhances diagnostic accuracy and operational efficiencies, supporting widespread adoption across urban and rural healthcare facilities.
Market Segmentation
Analysis by Test Type:
- Clinical Chemistry: Measures biomarkers and metabolic substances in specimens to assess organ function, detect disease, and monitor treatment with improved automation and high-throughput platforms.
- Molecular Diagnostics: Utilizes genetic material for disease detection at the molecular level, powered by PCR and NGS for precise early diagnoses of genetic, infectious, and cancerous conditions.
- Immunodiagnostics: Detects antibodies, antigens, or immune markers to diagnose infections, autoimmune diseases, and cancer with emerging biosensors and multiplex assays.
- Hematology: Studies blood cell counts, morphology, and clotting profiles for diagnosing anemia, leukemia, and other blood disorders with automation and data analysis enhancing accuracy.
- Others: Includes microbiology, point-of-care tests, and toxicology, benefiting from technological improvements in accuracy, speed, and usability in emergency and outpatient settings.
Analysis by Product:
- Reagents and Kits: Essential for testing infectious diseases, cancer, and genetic disorders with enhanced sensitivity, multiplexing, and faster, accurate results.
- Instruments: Includes analyzers, PCR machines, and automated systems that improve precision, throughput, and laboratory workflow efficiency.
Analysis by Usability:
- Disposable IVD Devices: Single-use products like test strips and sample kits, designed for point-of-care testing offering convenience and minimizing contamination.
- Reusable IVD Devices: Instruments like analyzers used repeatedly with cleaning and calibration, providing cost efficiency and supporting lab precision.
Analysis by Application:
- Infectious Disease: Detects pathogens (bacteria, viruses, fungi) using PCR, serology, and antigen tests for rapid, accurate results aiding timely treatment.
- Diabetes: Employs devices such as blood glucose meters, A1c tests, and continuous monitoring for individualized glycemic control.
- Cancer/Oncology: Includes assays like liquid biopsy and molecular profiling for early diagnosis, prognosis, and therapy monitoring.
- Cardiology: Utilizes biomarker assays including troponin, BNP, and lipid profiles to assess cardiovascular conditions and guide management.
- Autoimmune Disease: Detects antibodies or markers related to diseases like rheumatoid arthritis and lupus to support individualized treatment.
- Nephrology: Focuses on diagnosis of kidney diseases (CKD, AKI), aiding disease progression monitoring and treatment decision-making.
- Others: Covers conditions such as allergies, gastrointestinal, and neurological disorders, offering broad health monitoring solutions.
Analysis by End User:
- Hospitals: Provide comprehensive diagnostic services with advanced equipment for inpatients and outpatients.
- Laboratories: Offer routine and specialty diagnostics using cutting-edge technologies for accurate medical information.
- Clinical Laboratories: Facilitate precise diagnostics efficiently benefiting healthcare providers and patients.
- Point-of-care Testing Centers: Enable near-patient testing with immediate results for faster clinical decision-making.
- Academic Institutes: Drive diagnostics innovation via research, clinical studies, and training programs.
- Patients: Increasingly use home diagnostic tools for convenient monitoring of chronic conditions.
- Others: Include research organizations, public health agencies, and diagnostic companies applying IVD tools for epidemiological studies and product development.
Regional Insights
The Northeast region dominates the United States in vitro diagnostics market, with a concentration of major diagnostic companies, dense population, and top medical institutions. This region leads in growth for molecular diagnostics, oncology, and infectious disease testing. The Midwest follows with a growing market for immunodiagnostics and clinical chemistry, propelled by diverse healthcare settings. Meanwhile, the South sees higher healthcare spending, especially in cardiology, diabetes, and infectious diseases. The West is a biotechnology hub excelling in molecular diagnostics and autoimmune disease testing.
Recent Developments & News
In October 2024, Abbott Laboratories received authorization from the World Health Organization for its Alinity m MPXV assay for emergency use. This PCR-based diagnostic test detects mpox DNA from skin lesion swabs, enhancing testing capacity in outbreak regions and marking a key milestone under WHO’s Emergency Use Listing procedure.
Key Players
- Abbott
- Thermo Fisher Scientific Inc
- Agilent
- Hologic, Inc.
- Bio-Rad Laboratories
- QuidelOrtho Corporation
If you require any specific information that is not covered currently within the scope of the report, we will provide the same as a part of the customization.
Speak To an Analyst:
https://www.imarcgroup.com/request?type=report&id=43450&flag=C
Competitive Landscape:
The market research report offers an in-depth analysis of the competitive landscape, covering market structure, key player positioning, top winning strategies, a competitive dashboard, and a company evaluation quadrant. Additionally, detailed profiles of all major companies are included.
Key Highlights of the Report
1. Market Performance (2020-2025)
2. Market Outlook (2026-2034)
3. COVID-19 Impact on the Market
4. Porter’s Five Forces Analysis
5. Strategic Recommendations
6. Historical, Current and Future Market Trends
7. Market Drivers and Success Factors
8. SWOT Analysis
9. Structure of the Market
10. Value Chain Analysis
11. Comprehensive Mapping of the Competitive Landscape
About Us:
IMARC Group is a global management consulting firm that helps the world’s most ambitious changemakers create lasting impact. The firm offers comprehensive services for market entry and market expansion.
IMARC’s services include thorough market assessments, feasibility studies, company formation assistance, factory setup support, regulatory approvals and license navigation, branding, marketing and sales strategies, competitive landscape and benchmark analysis, pricing and cost studies, and sourcing studies.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No:(D) +91 120 433 0800
United States: +1-201971-6302
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- الألعاب
- Gardening
- Health
- الرئيسية
- Literature
- Music
- Networking
- أخرى
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness

