-
Fil d’actualités
- EXPLORER
-
Pages
-
Groupes
-
Evènements
-
Blogs
-
Marketplace
-
Forums
Biosimilars Market Opportunities for Pharma and Biotech Companies
The increasing role of biosimilars in hospital treatment protocols is another factor driving market growth. Hospitals are adopting biosimilars to manage healthcare budgets effectively. This shift is improving patient access to essential biologic therapies.
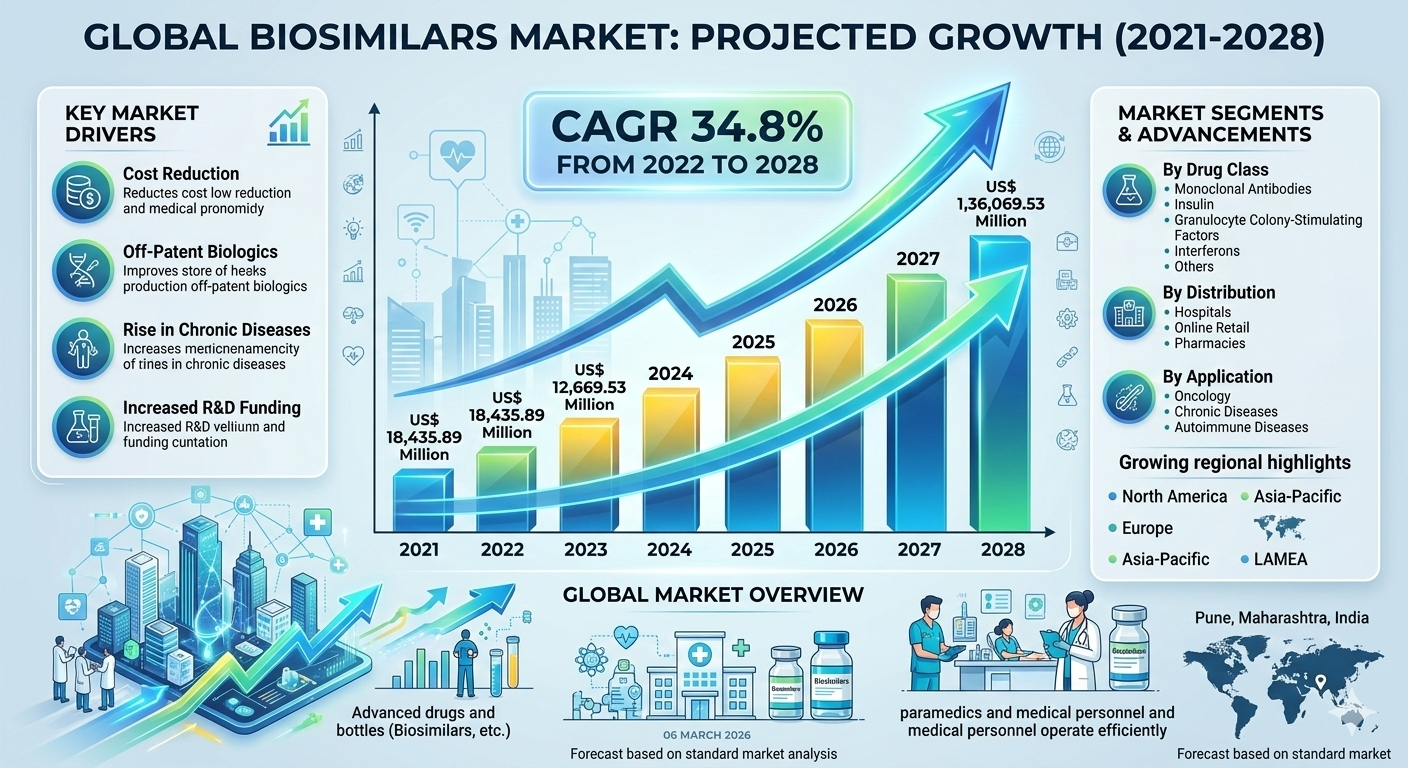
The global Biosimilars Market is experiencing remarkable growth as healthcare systems increasingly adopt cost-effective alternatives to biologic drugs. According to industry research, the biosimilars market size is projected to grow from US$ 18,435.89 million in 2021 to US$ 136,069.53 million by 2028, registering a strong CAGR of 34.8% during 2022–2028. The rising burden of chronic diseases and the increasing need for affordable treatment options are key factors supporting this rapid market expansion. Pharmaceutical companies are investing heavily in biosimilar drug development to address the growing demand for biologic therapies at lower costs.
Market Overview
Biosimilars are biological medicines that are highly similar to an already approved biologic drug, often referred to as the “reference product.” These medicines are developed to match the quality, safety, and effectiveness of the original biologic therapy. Regulatory authorities approve biosimilars under strict guidelines to ensure that they meet the same pharmaceutical standards as their reference drugs.
Biosimilars have become essential treatment options for several diseases, including cancer, autoimmune disorders, kidney conditions, and chronic inflammatory diseases. Conditions such as psoriasis, Crohn’s disease, rheumatoid arthritis, and diabetes require long-term treatment with biologic drugs, which are often expensive. Biosimilars provide a more affordable alternative while maintaining comparable clinical performance. This affordability is helping expand patient access to life-saving medications worldwide.
Download Sample PDF: https://www.theinsightpartners.com/sample/TIPHE100001246
Key Market Drivers
One of the major drivers of the biosimilars market is the increasing prevalence of chronic diseases. The global rise in cancer, autoimmune disorders, and metabolic diseases has created a strong demand for effective and affordable therapies. For instance, autoimmune conditions such as ankylosing spondylitis and rheumatoid arthritis continue to affect millions of patients worldwide, driving the need for long-term treatment solutions.
Biosimilars such as infliximab-based drugs are widely used to treat inflammatory and autoimmune diseases. According to research published in the Scandinavian Journal of Rheumatology, approximately 7.3% of the population in Spain tested positive for ankylosing spondylitis in screening studies. Such growing disease prevalence highlights the importance of biosimilars in providing accessible treatment options.
Another key factor driving market growth is the increasing number of regulatory approvals for biosimilar products. Regulatory agencies such as the U.S. Food and Drug Administration (FDA) are actively supporting the development and approval of biosimilar medicines. These approvals provide patients with more treatment options while encouraging pharmaceutical companies to invest in biosimilar research.
Increasing Product Approvals
In recent years, several biosimilar drugs have received regulatory approval, contributing to market expansion. Some notable approvals include Alymsys (bevacizumab-maly), Cimerli (ranibizumab-eqrn), Fylnetra (pegfilgrastim-pbbk), Vegzelma (bevacizumab-adcd), and Idacio (adalimumab-aacf). These biosimilars are developed as alternatives to well-known biologic drugs such as Avastin, Lucentis, Neulasta, and Humira.
The introduction of these products has significantly improved treatment accessibility while reducing healthcare costs. As more biosimilars enter the market, patients and healthcare providers gain additional treatment choices at competitive prices.
Regional Insights
North America represents one of the largest markets for biosimilars, driven by high healthcare spending, strong regulatory frameworks, and increasing adoption of biologic therapies. The North American biosimilars market was valued at US$ 5,479.84 million in 2021 and is expected to reach US$ 47,746.80 million by 2028, growing at a CAGR of 37.3%.
The United States holds the largest share of the regional market due to the growing prevalence of chronic diseases and the increasing number of biosimilar approvals. According to the National Institutes of Health (NIH), more than 24 million Americans suffer from autoimmune diseases, highlighting the strong demand for biologic and biosimilar treatments. Additionally, rising awareness among healthcare professionals and patients is supporting the adoption of biosimilars across the country.
Biosimilars also play an important role in reducing healthcare expenditure. Biologic drugs are among the most expensive medications, often costing thousands of dollars annually for a single patient. Biosimilars are typically priced 15% to 30% lower than their reference products, making them a cost-effective alternative. In the United States alone, biosimilars saved US$ 7.9 billion in healthcare costs in 2020, and this figure is expected to increase significantly as more biosimilars become available.
Competitive Landscape
The biosimilars market is highly competitive, with several global pharmaceutical companies actively developing and launching new products. Major companies operating in the market include Amgen Inc., Celltrion Inc., Sanofi SA, Biocon Ltd., Pfizer Inc., Samsung Bioepis Co. Ltd., Coherus BioSciences Inc., Eli Lilly and Company, Sandoz AG, Teva Pharmaceutical Industries Ltd., and Dr. Reddy’s Laboratories Ltd.
Among these companies, Pfizer and Novartis are considered leading players due to their strong biosimilar portfolios and global market presence. These organizations are continuously investing in research, partnerships, and product launches to strengthen their competitive positions.
Future Outlook
The future of the biosimilars market appears extremely promising as healthcare systems seek affordable alternatives to high-cost biologic drugs. Increasing regulatory support, rising chronic disease prevalence, and continuous technological advancements in biotechnology are expected to drive long-term market growth. As more biosimilars receive approval and enter the global market, they will play a critical role in improving healthcare accessibility and reducing treatment costs for patients worldwide.
Also Available in :
Korean German Japanese French Chinese Italian Spanish
- biosimilars_market
- biosimilars_market_growth
- biosimilars_market_size
- biosimilars_market_forecast
- global_biosimilars_market
- biosimilar_drugs_market
- biosimilars_pharmaceutical_industry
- biosimilars_industry_trends
- biosimilars_market_analysis
- biologics_and_biosimilars_market
- biosimilar_drug_development
- biosimilars_market_demand
- biosimilar_therapeutics_market
- biosimilars_market_opportunities
- biosimilars_market_CAGR
- biosimilars_healthcare_market
- biosimilars_industry_outlook
- biosimilars_regulatory_landscape
- biosimilars_competition_market
- biosimilars_pharmaceutical_research
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jeux
- Gardening
- Health
- Domicile
- Literature
- Music
- Networking
- Autre
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness

