-
Nieuws Feed
- EXPLORE
-
Pagina
-
Groepen
-
Events
-
Blogs
-
Marketplace
-
Forums
Pharmacovigilance Outsourcing Market: Navigating the 16.1% CAGR Growth Trajectory (2025–2031)
The global pharmacovigilance outsourcing market healthcare landscape is witnessing a paradigm shift in how drug safety and regulatory compliance are managed. As pharmaceutical and biotechnology companies face mounting pressure to accelerate time to market while adhering to increasingly stringent safety standards, the Pharmacovigilance (PV) Outsourcing Market has emerged as a critical strategic pillar. This sector involves the delegation of drug safety activities, such as adverse event reporting, case processing, and signal detection, to specialized third party service providers. By leveraging external expertise, life sciences organizations can focus on their core competencies of drug discovery and development while ensuring patient safety is maintained at the highest level.
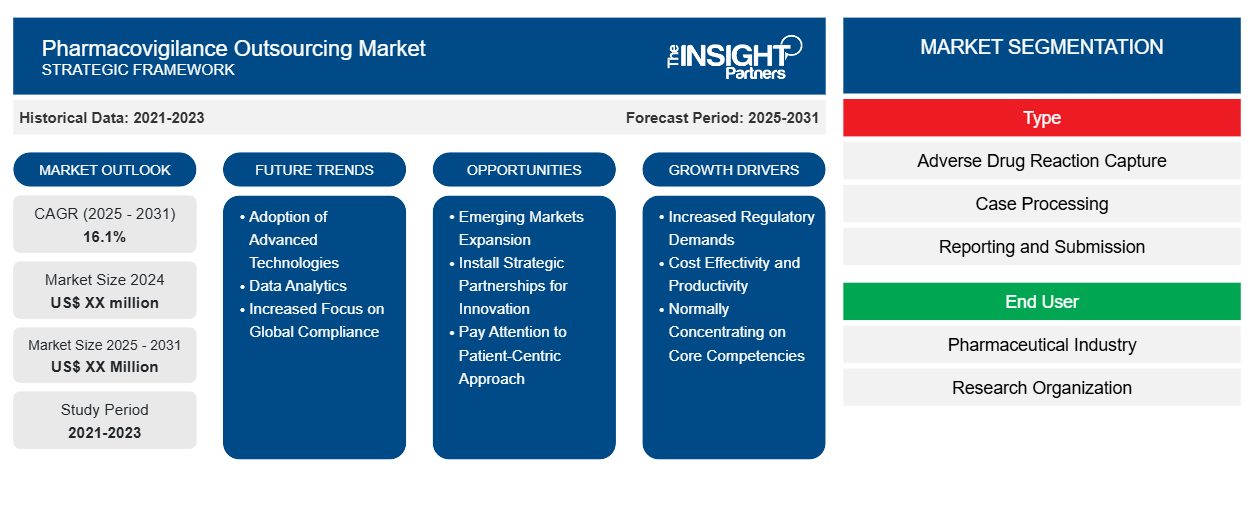
The is poised for significant expansion in the coming years. According to recent industry analysis, the market is expected to register a CAGR of 16.1% from 2025 to 2031. This robust growth rate reflects a broader trend toward cost optimization and the adoption of advanced technology in clinical and post marketing surveillance. As the volume of safety data continues to explode due to the rise in clinical trials and the proliferation of complex biologics, the reliance on Contract Research Organizations (CROs) and Business Process Outsourcing (BPO) firms is becoming more of a necessity than a choice.
Download Sample Report - https://www.theinsightpartners.com/sample/TIPRE00040657
Key Market Report Drivers
The impressive growth of the Pharmacovigilance Outsourcing Market is fueled by several interconnected drivers that are reshaping the industry.
1. Escalating Volume of Adverse Drug Reaction (ADR) Reporting
One of the primary catalysts for market expansion is the rising incidence of adverse drug reactions globally. As the global population ages and the prevalence of chronic diseases increases, the consumption of pharmaceutical products has surged. This has naturally led to a higher volume of safety data that must be processed, analyzed, and reported to regulatory bodies like the FDA and EMA. Outsourcing provides the scalability required to handle these massive data sets without the need for significant capital investment in in-house infrastructure.
2. Stringent and Evolving Regulatory Requirements
Regulatory authorities worldwide are continuously updating their safety guidelines to enhance patient protection. Compliance is no longer a static goal but a dynamic process that requires constant monitoring of new mandates, such as the EU’s Medical Device Regulation (MDR) or updated GVP (Good Pharmacovigilance Practices) modules. For many small to mid-sized biotech firms, keeping pace with these shifting regulations is a monumental task. Outsourcing partners bring specialized regulatory intelligence and localized expertise, ensuring that companies remain compliant across multiple jurisdictions.
3. Rising Research and Development (R&D) Investments
The pharmaceutical industry is characterized by high R&D spending, particularly in therapeutic areas like oncology, immunology, and neurology. With more drugs entering clinical trial phases, the demand for pre-marketing pharmacovigilance services has skyrocketed. Outsourcing allows sponsors to manage the safety profiles of experimental drugs efficiently, providing a seamless transition from clinical trials to post-market surveillance.
4. Technological Advancements: AI and Automation
The integration of Artificial Intelligence (AI), Machine Learning (ML), and robotic process automation (RPA) into PV workflows is a major trend. Advanced outsourcing providers are utilizing AI to automate routine tasks like case intake and narrative writing. These technologies not only reduce human error but also significantly lower operational costs. The shift from manual to automated PV processes is a significant factor driving companies to seek out tech-enabled service providers.
Market Segmentation and Regional Outlook
The market is generally segmented by service type, service provider, and end user. Currently, post-marketing services hold a dominant share of the market, driven by the continuous monitoring required for drugs already on the shelves. However, pre-marketing services are expected to grow rapidly as the number of molecules in the clinical pipeline increases.
From a regional perspective, North America continues to lead the market due to its well-established pharmaceutical sector and the presence of major industry players. However, the Asia Pacific region is anticipated to witness the highest growth rate during the forecast period. Factors such as lower operational costs, a growing number of clinical trials in countries like India and China, and an increasing focus on healthcare infrastructure make APAC an attractive destination for PV outsourcing.
Strategic Importance of Top Industry Players
The competitive landscape of the Pharmacovigilance Outsourcing Market is characterized by the presence of several global leaders who are consistently innovating to provide end-to-end safety solutions. These players offer a blend of clinical expertise and technological prowess, helping sponsors navigate the complexities of global drug safety.
Top players in the market include:
- Accenture PLC: Known for its large-scale BPO services and digital transformation capabilities.
- IQVIA Holdings Inc.: A leader in clinical research and data analytics, providing integrated PV solutions.
- ICON plc: Specialized in clinical trial management and comprehensive safety reporting.
- Cognizant Technology Solutions: Focuses on leveraging AI and automation to streamline safety operations.
- Labcorp Drug Development (formerly Covance): Offers extensive laboratory and clinical safety services.
- Genpact Limited: A major player in process excellence and risk management services.
- Capgemini: Provides robust IT and business process solutions for the life sciences sector.
- Ergomed plc: Recognized for its niche expertise in specialized therapeutic areas.
Future Outlook: A Tech-Driven Safety Ecosystem
Looking ahead, the Pharmacovigilance Outsourcing Market is moving toward a more proactive and predictive model. The focus is shifting from simple compliance to "safety intelligence," where data is used not just to report events but to predict potential risks before they manifest. The use of Real World Evidence (RWE) and social media monitoring are becoming increasingly important components of a comprehensive PV strategy.
As companies continue to face the dual challenges of cost containment and regulatory pressure, the outsourcing model will remain indispensable. The projected 16.1% CAGR underscores the industry’s confidence in external partners to deliver high-quality, compliant, and cost-effective drug safety services. In this evolving environment, the synergy between pharmaceutical innovators and specialized PV providers will be the cornerstone of global patient safety and public health.
Related Report :
· Pharmacovigilance Market Dynamics, Analysis, Trends, and Opportunities 2031
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Spellen
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness

