-
Noticias Feed
- EXPLORE
-
Páginas
-
Grupos
-
Eventos
-
Blogs
-
Marketplace
-
Foros
Medical Device Testing and Certification Market Share Analysis and Competitive Landscape
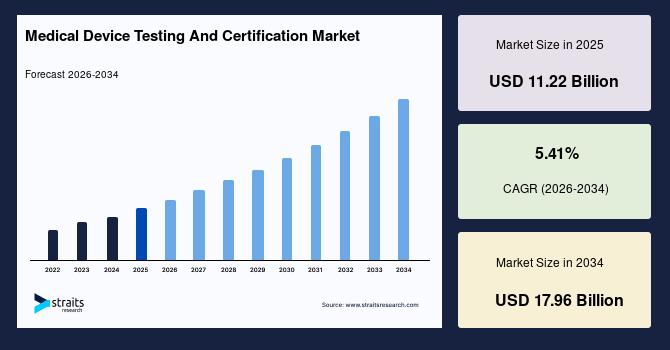
The global Medical Device Testing and Certification Market is witnessing steady growth due to increasing regulatory requirements, rising demand for safe and high-quality medical devices, and continuous technological advancements in healthcare equipment. According to the latest report by Straits Research, the market was valued at USD 11.22 billion in 2025 and is projected to grow from USD 11.78 billion in 2026 to reach USD 17.96 billion by 2034, registering a CAGR of 5.41% during the forecast period. The increasing adoption of AI-enabled medical devices, stringent global compliance standards, and growing focus on patient safety are major factors driving market expansion.
Market Drivers
Increasing Regulatory Requirements Boost Market Growth
The medical device testing and certification market is primarily driven by the increasing implementation of stringent regulatory frameworks across the healthcare industry. Regulatory authorities such as the FDA and the European Commission are enforcing strict compliance requirements to ensure device safety, quality, and performance. These regulations are increasing the demand for testing, inspection, and certification services among medical device manufacturers.
Rising Adoption of Advanced Medical Technologies
Another major driver is the rapid advancement of medical technologies. The growing use of connected healthcare devices, AI-powered diagnostic systems, wearable devices, and implantable medical technologies has significantly increased the complexity of medical devices. As a result, manufacturers require comprehensive testing and certification services to validate device performance, cybersecurity, functionality, and compliance with international standards.
Growing Healthcare Investments and Chronic Disease Prevalence
The rising prevalence of chronic diseases and increasing healthcare investments are also supporting market growth. Demand for advanced diagnostic equipment, monitoring systems, and therapeutic devices continues to rise globally, creating a strong need for reliable testing and certification solutions before product commercialization.
Expansion of Medical Device Single Audit Program (MDSAP)
In addition, the increasing adoption of the Medical Device Single Audit Program (MDSAP) is streamlining international approvals and encouraging manufacturers to invest in unified compliance procedures. The growing emphasis on lifecycle assurance and post-market surveillance is further boosting demand for continuous testing and certification services.
Market Challenges
High Cost of Testing and Certification Procedures
Despite strong growth potential, the market faces several challenges. One of the primary restraints is the high cost associated with testing and certification procedures. Advanced medical devices require extensive validation, clinical testing, documentation, and regulatory approvals, which can significantly increase operational costs for manufacturers.
Complex and Evolving Regulatory Landscape
Another challenge is the constantly evolving regulatory environment. Frequent updates in global medical device regulations create compliance complexities and increase approval timelines for companies operating across multiple regions. Small and medium-sized manufacturers often face difficulties managing changing compliance requirements and maintaining certification standards.
Shortage of Skilled Regulatory Professionals
Additionally, the shortage of skilled regulatory professionals and technical experts can hinder efficient testing and approval processes. Delays in certification procedures and supply chain disruptions may also affect market growth.
Market Segmentation
By Service Type
The market is segmented into testing services, inspection services, and certification services. The testing services segment dominates the market due to increasing demand for safety validation, performance testing, microbiological testing, and biocompatibility assessments for medical devices.
By Sourcing Type
Based on sourcing type, the market is divided into in-house and outsourced services. The in-house segment holds a significant market share as large medical device manufacturers prefer internal compliance management and testing infrastructure. However, outsourced testing services are also gaining popularity among small and mid-sized manufacturers seeking cost-effective compliance solutions.
By Device Class
The market includes Class I, Class II, and Class III devices. The Class II segment accounts for a major market share due to the widespread use of diagnostic and therapeutic devices requiring moderate regulatory control and testing procedures. The Class III segment is expected to witness strong growth due to rising demand for implantable and life-supporting medical devices.
By Technology
The market is segmented into active implant medical devices, active medical devices, non-active medical devices, in-vitro diagnostic devices, ophthalmic medical devices, orthopedic and dental medical devices, vascular medical devices, and others. Non-active medical devices currently dominate the market because of their extensive usage in hospitals and healthcare facilities worldwide.
By End-User
The market primarily caters to medical device manufacturers, healthcare institutions, and regulatory organizations. Medical device manufacturers account for the largest market share due to increasing investments in regulatory compliance and product quality assurance.
Regional Insights
North America Dominates the Global Market
North America accounted for the largest market share in 2025. The region’s growth is supported by advanced healthcare infrastructure, strong regulatory frameworks, and the presence of major medical device manufacturers in the United States and Canada. Increasing FDA approvals and growing adoption of AI-enabled medical devices are further boosting regional demand.
Europe Witnesses Strong Growth
Europe is expected to witness the fastest growth during the forecast period. The implementation of the European Union Medical Device Regulation (EU MDR) and increasing recertification requirements for existing devices are significantly driving demand for testing and certification services across the region.
Asia-Pacific Emerging as a Key Growth Region
Asia-Pacific is also emerging as a rapidly growing market due to rising healthcare investments, expanding medical device manufacturing activities, and increasing regulatory awareness in countries such as China, India, and Japan. Growing exports of medical devices from Asia-Pacific countries are creating substantial opportunities for certification service providers.
Growth Opportunities in Latin America and Middle East & Africa
Latin America and the Middle East & Africa are witnessing gradual growth due to improving healthcare infrastructure, increasing medical device imports, and growing emphasis on regulatory compliance and patient safety.
Key Players Analysis
Major Companies Focus on Expansion and Innovation
The Medical Device Testing and Certification Market is highly competitive, with companies focusing on strategic partnerships, technological advancements, acquisitions, and service expansion to strengthen their market presence.
Key players operating in the market include Charles River Laboratories, TÜV SÜD, Intertek, Accuprec Research Labs, American Preclinical Services, SGS SA, Bureau Veritas, TÜV Rheinland, UL LLC, Eurofins Scientific, Element Materials Technology, and WuXi AppTec.
These companies are investing heavily in advanced testing technologies, regulatory consulting services, and global laboratory expansion to meet increasing compliance requirements and improve customer support capabilities. Strategic collaborations with medical device manufacturers are also helping companies enhance their competitive positioning in the global market.
Refer : https://straitsresearch.com/report/medical-device-testing-and-certification-market
About Us
Straits Research is a leading research and intelligence organization, specializing in research, analytics, and advisory services, along with providing business insights and research reports.
Contact Us
Email: sales@straitsresearch.com
Tel: +1 646 905 0080 (U.S.), +44 203 695 0070 (U.K.)
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Juegos
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness

